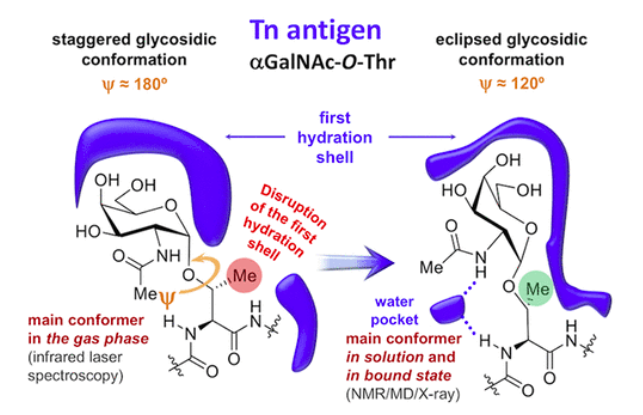
We report that two seemingly similar variants of Tn antigens have very different conformations in water. Using interdisciplinary approaches, both experimental and computational, the team showed that the threonine-linked Tn antigen assumes a rigid form in solution and forms a persistent water pocket. In contrast, the serine-linked Tn antigen lacks the water pocket and is flexible in solution. These differences were not observed in gas phase studies.
The different Tn antigen conformations are likely to result in distinct interactions with cellular receptors and antibodies and understanding these structures may facilitate the design of more effective screening tools and anticancer drugs.
The investigation has been published in JACS.
 Spectroscopy Group of Basque Country University Structural study of biological molecules in the gas phase using Microwave Spectroscopy
Spectroscopy Group of Basque Country University Structural study of biological molecules in the gas phase using Microwave Spectroscopy


